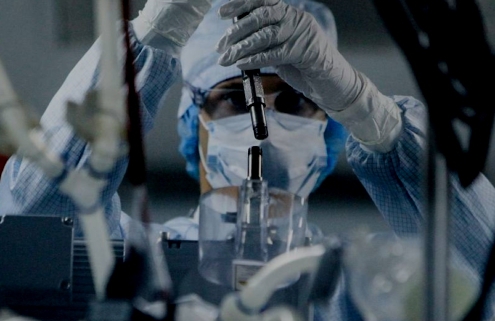
X7 Research offers a wide range of services in all phases of building a pharmacovigilance system, from individual consultations to complete pharmacovigilance.
SOCIAL RESPONSIBILITY
X7 Research’s primary responsibility is to help our clients in bringing medicines & products to the market efficiently and safely.
The Best CRO for Clinical Trials | Full-service Global CRO — X7 Research
The Best CRO in Pharma | Clinical Research Organisation — X7 Research
The Best Contract Research Organisation (CRO) | Trusted CRO — X7 Research
The Best Clinical Research Organisation (CRO): Full Service CRO | X7 Research
The Best CRO in Research | Trusted CRO for Your Trail — X7 Research
The Best Clinical Trial Company | Global Full-service CRO - X7 Research
The Best Pharma CROs: Clinical Trials for Drugs | X7 Research
The Best CROs Company: Clinical Trials with Global CRO — X7 Research
The Best Pre-clinical CRO: Safety Assessment With X7 Research
The Best Clinical Development | Trusted CRO for Your Product: X7 Research
The Best Biostatistical Consulting: Statistical Services | X7 Research
The Best Pharmacovigilance Consulting Services | CRO — X7 Research
Feasibility in Clinical Trials: What is a Feasibility in Clinical Trials?