First-of-a-kind malaria vaccine
GlaxSmithKline announced the launch of clinical trials of Mosquirix, malaria vaccine, first-of-a-kind, approved by World Health Organization. This project is pilot, but WHOs managers are sure of results will provide the data for enlarging future studies.
Clinical trials of vaccine will be conducted during two years with the participation of infected babies from Malawi, Ghana, and Kenia, about 360 thousand subjects should be covered. “Combined with existing malaria interventions, such a vaccine would have the potential to save tens of thousands of lives in Africa,” said Matshidiso Moeti, WHO regional director for Africa.
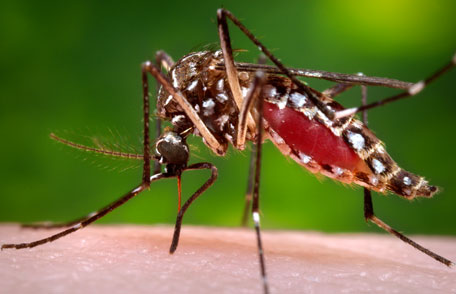
Malaria high rates were the reason of chose these three countries, despite broad use of insecticidal bed nets (malaria is transferred with infected insect bite) and well-functioning immunization programs. It is up to countries to decide, which district should be included in trials, nevertheless, the priority is given to malaria-heavy areas.
The international vaccine alliance Gavi is providing $27.5 million for the tests. Another $15 million comes from the Global Fund to Fight AIDS, Tuberculosis, and Malaria. With $9.6 million more from UNITAID, WHO has secured all funding for the pilot studies. The work could take four to five years to complete.
GSK developed the vaccine with help from the PATH Malaria Vaccine Initiative and with financial support from the Bill and Melinda Gates Foundation.