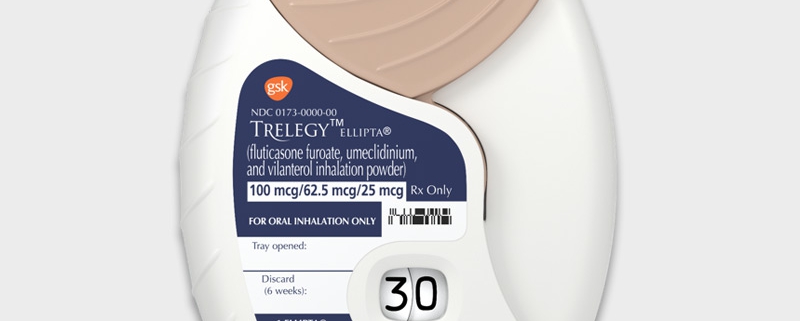
Trelegy – a new drug from GSK
Facing the prospect of generic competition to respiratory superstar Advair—and as the blockbuster’s sales falter under pricing pressure—GlaxoSmithKline is pushing to grow new drug Trelegy Ellipta by seeking an expanded indication at the FDA.
Along with partner Innoviva, GSK applied for approval for Trelegy as a maintenance treatment of airflow obstruction and to reduce exacerbations for patients with chronic obstructive pulmonary disease (COPD).
Trelegy Ellipta is the first FDA-approved closed triple drug that puts three COPD drugs in one inhaler: a LABA, a LAMA and a steroid. GSK’s drug contains vilanterol, umeclidinium and fluticasone furoate. Before the approval and launch, patients who needed all three drugs had to use more than one inhaler, or an open triple treatment.
Trelegy is one of a group of recently approved drugs. GSK hopes that this will help overcome the loss of Advair’s position. On the company’s third-quarter conference call, GSK CFO Simon Dingemans said Trelegy is a “key addition to the Ellipta portfolio, and one which we believe will be a significant new growth driver for the respiratory business.” However, he warned that it would take time to build sales, as the drug only received the FDA’s first approval in September.
Elsewhere in GSK’s portfolio, new shingles vaccine Shingrix picked up approval in October. Dingemans said on the conference call the company thinks “these two new products represent significant innovation that will benefit patients, and we remain confident in their long-term significance for the group.”
Just recently, a new HIV combination from GSK was supported by the FDA. Together, the three new preparations for launch are a near-term focus for GSK, as the company tries to overcome competition on all fronts, CEO Emma Walmsley has told analysts and investors since coming on board earlier this year.
Source: fiercepharma.